Zoliflodacin: A Comprehensive Review of a First-in-Class Spiropyrimidinetrione for the Treatment of Multi-Drug-Resistant Neisseria gonorrhoeae
JASPI March 2026 / Volume 4 /Issue 1
Copyright: © Author(s). This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Nayak BS, C M, Rangari GM, et al.Zoliflodacin: A Comprehensive Review of a First-in-Class Spiropyrimidinetrione for the Treatment of Multi-Drug-Resistant Neisseria gonorrhoeae. JASPI. 2026;4(1):20-29 DOI: 10.62541/jaspi126
ABSTRACT
KEYWORDS: Zoliflodacin; ETX0914; Neisseria Gonorrhoeae; Spiropyrimidinetrione; Anti-microbial Resistance
INTRODUCTION
Gonorrhoea, caused by the bacterium Neisseria gonorrhoeae, poses a significant global health challenge.1 This pathogen has developed resistance to multiple antimicrobials, including penicillins and tetracyclines, and is increasingly resistant to ceftriaxone, the primary treatment of Gonorrhoeae.2 The World Health Organization has labelled Neisseria gonorrhoeae a high-priority pathogen for antimicrobial research and development that led to development of novel agents like Zoliflodacin.2 Antimicrobial-resistant gonorrhoea is emerging and necessitates new treatment options. Zoliflodacin is a novel Spiropyrimidinetrione antibiotic developed to combat multidrug-resistant N. gonorrhoeae strains.3,1 With support from the Global Antibiotic Research and Development Partnership (GARDP), it has advanced through later clinical phases.4
Substantial resistance to various antimicrobials previously used in the treatment of gonorrhoea, including fluoroquinolones, macrolides, and oral cephalosporins, was confirmed from surveillance data conducted in multiple regions.5 The presence of multidrug-resistant N. gonorrhoeae isolates has also been evidenced on a global scale, further limiting treatment options.6 These data highlight the need to examine novel agents with activity against resistant strains and distinct mechanisms of action, such as Zoliflodacin.1 This review summarizes and critically evaluates the available evidence on zoliflodacin, including its mechanism of action, antimicrobial activity, pharmacokinetics and pharmacodynamics, clinical efficacy, safety profile, and resistance patterns.
BACKGROUND & MECHANISM OF ACTION
Zoliflodacin has antibacterial effect by inhibiting bacterial enzyme type II topoisomerases via a novel mechanism of action that targets DNA gyrase subunit B (GyrB).7, 4 In contrast to fluoroquinolones (which bind and act mainly on the GyrA subunit and require a water-metal ion bridge for binding and acting), Zoliflodacin operates by binding directly to the highly conserved residues of GyrB, hinting at a potentially more durable mechanism that may be less vulnerable to resistance development.4
Zoliflodacin binds at the same site of DNA cleavage as quinolones, but uses a different mechanism of binding4. This stabilizes DNA cleavage complexes with DNA gyrase, sterically blocking DNA religation and thereby inhibiting bacterial DNA biosynthesis.4 Importantly, Zoliflodacin binding to some of the most conserved GyrB residues, regardless of the quinolone water-metal ion bridge to GyrA, indicates that bacteria probably have a harder time developing target-mediated resistance via mutations.4
Notably, Zoliflodacin displayed more efficacy against fluoroquinolone-resistant gyrase than against topoisomerase IV, showing that the drug can overcome existing quinolone resistance mechanisms (Figure 1).8 The spiropyrimidinetrione scaffold is an entirely different chemical class from quinolones, offering a different pharmacophore for bacterial topoisomerase inhibition.3,1
Figure 1: Mechanism of action of Fluoroquinolones and Zoliflodacin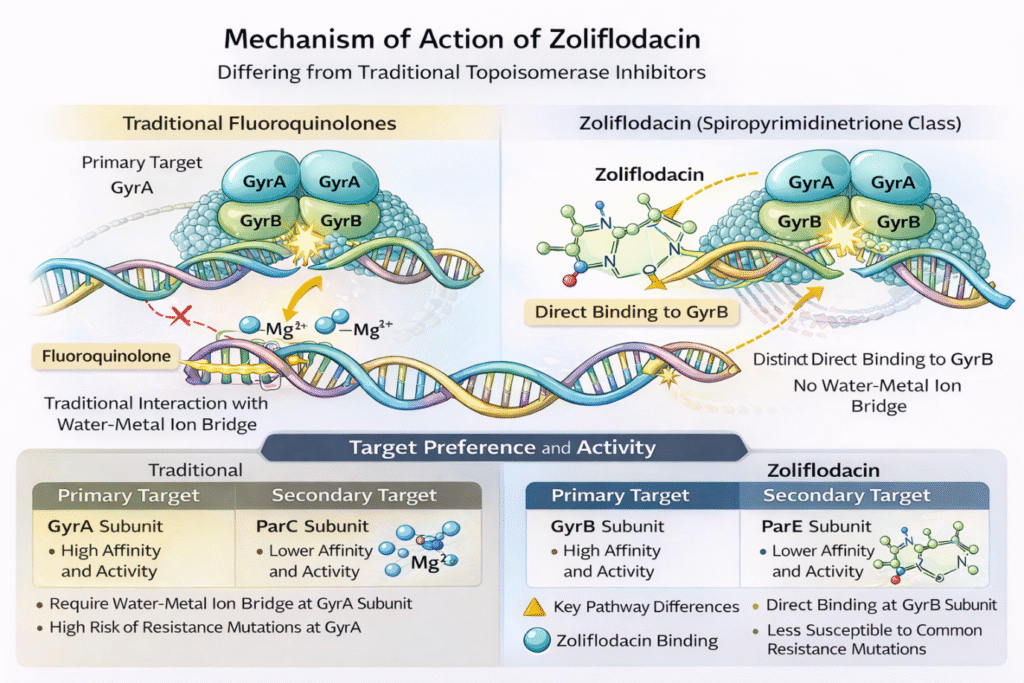
ANTIMICROBIAL ACTIVITY AND SPECTRUM
Zoliflodacin has very good in vitro activity against N. gonorrhoeae isolates from many geographic regions and with different resistance backgrounds. Notably, cross-resistance with antimicrobials used today or before for gonorrhoea treatment, including cephalosporins, macrolides, and fluoroquinolones, has not been reported.9
Zoliflodacin has been widely validated against modern clinical isolates in large-scale surveillance studies. Importantly, Zoliflodacin did not show cross-resistance with other tested antimicrobials during its investigation, and targeted GyrB was highly conserved, with no isolates with markedly elevated MIC values suggestive of reduced susceptibility were identified.5 Importantly, no cross-resistance was observed between Zoliflodacin and ciprofloxacin, although both drugs inhibit DNA topoisomerase II enzymes.10
Global surveillance across Europe, China, and South Korea confirms that Zoliflodacin maintains potent in-vitro activity against both wild-type isolates and isolates resistant to other antimicrobial classes, with MIC values distributed within a relatively narrow range across geographically diverse datasets. Detailed regional MIC distributions are summarised in Table 1
Table-1: Zoliflodacin MIC data across geographical regions
|
Region |
Sample Size |
Study Year |
MIC₅₀ (µg/mL) |
MIC₉₀ (µg/mL) |
MIC Range |
Reference |
|
Europe (25 countries) |
1,209 |
2018 |
0.125 |
0.25 |
0.03- |
Unemo et al., 2021 |
|
China (Nanjing) |
200 |
2014- |
0.06 |
0.125 |
0.03- |
Le et al., 2021 |
|
South Korea |
150 |
2024 |
0.03 |
0.06 |
0.015-0.125 |
Roh et al., 2025 |
|
United States |
180 |
2019- |
0.08 |
0.125 |
0.03- |
Scangarella-Oman et al., 2020 |
|
Global (diverse) |
71 |
2018- |
0.06 |
0.125 |
0.03- |
Golparian et al., 2022 |
|
MDR isolates (China) |
200 |
2014- |
0.06 |
0.125 |
0.03- |
Le et al., 2021 |
Note: MIC₅₀ and MIC₉₀ represent the concentrations inhibiting 50% and 90% of isolates, respectively, and describe population-level in vitro activity rather than formal susceptibility categorization in the absence of established clinical breakpoints
Clinical susceptibility breakpoints for Zoliflodacin have not yet been formally established by CLSI or EUCAST; therefore, MIC data are presented descriptively to illustrate observed distributions rather than categorical susceptible or resistant classifications.
Longitudinal screening by Nanjing, China, with 986 isolates acquired during 2014–2018, showed overall MIC50 and MIC90 of 0.06 mg/L and 0.125 mg/L, respectively.6 Although a modest temporal rightward shift in MIC distributions was observed between 2014 and 2018, MIC values remained within the previously reported distribution range, including among isolates resistant to ciprofloxacin, azithromycin, and extended-spectrum cephalosporins.6
Other than N. gonorrhoeae, evidence suggests that Zoliflodacin may be active against other bacterial pathogens. The MIC of the drug was 4 μg/mL against Acinetobacter baumannii, a 4-fold enhanced activity against the progenitor compound QPT-1.4 In addition, Zoliflodacin demonstrates in vitro activity against Helicobacter pylori isolates with MIC values between 0.008 and 1 μg/mL (MIC50: 0.125 μg/mL; MIC90: 0.25 μg/mL), as well as with no cross-resistance to first-line antibiotics.11 Though clinical developments have been conducted mainly with gonococcal infections, these results point toward potential applications beyond gonorrhoea. The clinical relevance of these MIC distributions is supported by pharmacokinetic – pharmacodynamic modelling and confirmed by Phase 3 clinical trial outcomes, rather than by breakpoint-based susceptibility interpretation.
PHARMACOKINETICS AND PHARMACODYNAMICS
Phase 1 studies of Zoliflodacin have well characterized its pharmacokinetics in healthy volunteers. After oral administration, Zoliflodacin is rapidly absorbed, with a time to maximum concentration (Tmax) of between 1.5 and 2.3 hours in the fasted state.12 Drug exposure increases dose-proportionally up to 800 mg, supporting predictable pharmacokinetics across the therapeutic dose range.12
Metabolism and excretion studies with radiolabelled Zoliflodacin confirmed that the predominant pathway of elimination was metabolism and faecal excretion (79.6%), with minimal renal clearance of the unchanged drug (2.5%)11. Following a single 3 g oral dose, Zoliflodacin is rapidly absorbed with a mean time to maximum concentration (Tmax) of 2.5 ± 0.8 hours in the fasted state, which is not significantly altered by food intake.
The peak plasma concentration (Cmax) was
38 ± 8 µg/mL and with sustained exposure below equation:
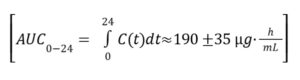
Above written formula resulted in AUC/MIC ratio of approximately 1,520, greatly exceeding the known efficacy thresholds for N. gonorrhoeae. The drug has a moderate volume of distribution (85 ± 15L) and a terminal half-life of 6.0 ± 1.2 hours, forming a pharmacokinetic profile that is highly favourable for use as a single-dose oral therapy. Elimination is by means of the biliary-fecal pathway (79.6%), with minimal renal excretion (2.5%), indicating an acceptable safety profile in patients with renal impairment
Population pharmacokinetic (PPK) analyses using information from 261 subjects from six phase 1 studies and 24 patients in the pivotal phase 3 trial reported numerous key factors as determinants of pharmacokinetic variability. The final PPK model consisted of an oral depot compartment, a transit chain for absorption, and one systemic compartment. Body weight and co-administration of strong CYP3A4 inhibitors were significant predictors of pharmacokinetic parameters.
Data from pharmacodynamic studies confirmed that the choice of a single oral dose of 3 g was suitable for uncomplicated gonorrhoea. With non-clinical PK-PD and MIC in the PPK model, the probability of target attainment (PTA) reached ≥96.2% with MIC ≤0.25 μg/mL, encompassing 99.6-100% of clinical isolates. By averaging over MIC distributions from surveillance studies, PTA was ≥99.5%, which provided high confidence for this dose regimen.
Dynamic hollow fibre infection model (HFIM) studies have elucidated the pharmacodynamic process. Regarding Zoliflodacin-susceptible strains, single oral doses of 3 g and 4 g successfully eradicated N. gonorrhoeae, with no growth recovered during 7-day experiments 12. However, lower doses (0.5 g, 1 g, and 2 g) failed to eradicate bacteria and selected for resistant populations with GyrB D429N substitutions 12. For strains harboring the GyrB S467N substitution conferring reduced susceptibility, doses ≥3 g were required for effegonorrhea.ent 12.
CLINICAL TRIALS AND EFFICACY
Zoliflodacin has been promoted through well-rounded clinical development to crucial Phase 3 trials. The first Phase 2 trial provided proof of concept for single-dose therapy for the treatment of urogenital gonorrhoea using Zoliflodacin, establishing both the basis for dose selection and the refinement of the compound13.
Phase 3 Clinical Trial Results
In the pivotal international, randomized, open-label, non-inferiority phase 3 trial, a single 3 g oral dose of Zoliflodacin was compared with intramuscular ceftriaxone (500 mg) plus oral azithromycin (1 g) for treatment of uncomplicated urogenital gonorrhoeae. A total of 930 participants were randomized (2:1), and 927 received treatment and were included in the safety population. The microbiological intention-to-treat (urogenital) population included 744 participants with culture-confirmed baseline infection.14
The primary endpoint was microbiological cure at the urogenital site at the test-of-cure visit (day 6 ± 2). Cure rates in the microbiological intention-to-treat population were 90.9% (460/506) in the Zoliflodacin group and 96.2% (229/238) in the comparator group, yielding a treatment difference of 5.3% (95% CI 1.4–8.6). The upper bound of the confidence interval was below the prespecified non-inferiority margin of 12%, thereby meeting criteria for non-inferiority.14
In the evaluable population, which excluded participants with non-assessable test-of-cure outcomes, microbiological cure rates increased to 96.8% (460/475) with Zoliflodacin and 100% (229/229) with comparator therapy. Sensitivity analyses using alternative assumptions for missing data produced consistent results, supporting the robustness of the primary endpoint analysis.14
Extragenital outcomes were analyzed as secondary endpoints. In the microbiological intention-to-treat population, rectal cure rates were 87.3% (69/79) for Zoliflodacin and 88.6% (31/35) for comparator therapy. Pharyngeal cure rates were 79.2% (42/53) and 78.6% (22/28), respectively. The study was not powered to establish non-inferiority at extragenital anatomical sites, and participant numbers in these subgroups were limited.14
Both treatments were generally well tolerated. Treatment-emergent adverse events occurred in 46% of participants in each group. The most frequently reported adverse events in the Zoliflodacin group were headache (10%), neutropenia (7%), and leukopenia (4%), whereas injection-site pain (12%), neutropenia (8%), and diarrhoea (7%) were most common in the comparator group. No serious adverse events or treatment discontinuations due to adverse events were reported.14
The predefined non-inferiority margin was 12%. The observed treatment difference of 5.3% with an upper 95% confidence limit of 8.6% remained within this margin, confirming statistical non-inferiority. Although cure rates numerically favoured comparator therapy, confidence intervals did not cross the prespecified threshold, supporting the primary endpoint conclusion. Sensitivity analyses and evaluable population results demonstrated consistent findings, strengthening internal validity. This result interpretation was summarized in Table.2
Table 2: Phase-3 Clinical trial efficacy and safety outcomes
|
Outcome |
Zoliflodacin 3 g |
Ceftriaxone + Azithromycin |
Difference (95% CI) |
|
Primary endpoint (urogenital cure, ITT) |
90.9% (460/506) |
96.2% (229/238) |
5.3% (1.4–8.6) |
|
Evaluable population |
96.8% (460/475) |
100% (229/229) |
3.2% (1.1–5.1) |
|
Rectal cure (ITT) |
87.3% (69/79) |
88.6% (31/35) |
— |
|
Pharyngeal cure (ITT) |
79.2% (42/53) |
78.6% (22/28) |
— |
|
Any adverse event |
46% |
46% |
— |
|
Serious adverse events |
0 |
0 |
— |
ITT =Microbiological Intention-to-treat population
Analyses were conducted in the microbiological intention-to-treat population, with participants lacking assessable test-of-cure results classified as treatment failures. A secondary evaluable population analysis excluded protocol deviations and missing outcomes. Non-inferiority was concluded if the upper bound of the two-sided 95% confidence interval for the treatment difference remained below the prespecified margin of 12%.
Taken together, these findings demonstrate that single-dose oral Zoliflodacin achieved microbiological cure rates consistent with non-inferiority to standard dual therapy while maintaining a comparable safety profile. Although extragenital efficacy requires further investigation in adequately powered studies, the results support Zoliflodacin as a clinically viable oral alternative for uncomplicated urogenital gonorrhoeae.
Pharmacodynamic modelling using hollow fibre infection models has further supported clinical effectiveness. Combination therapy with Zoliflodacin plus doxycycline was also marginally more effective than Zoliflodacin monotherapy in patients with concomitant chlamydial infection15. The combination eradicated susceptible strains with a 0.5 g single dose of Zoliflodacin plus doxycycline 200 mg daily for 7 days and eradicated extensively drug-resistant strains with a 2 g Zoliflodacin dose plus doxycycline15. Crucially, the combined approach effectively suppressed the emergence of Zoliflodacin resistance more effectively than monotherapy15.
The clinical development program has primarily focused on urogenital gonorrhoea, with less extensive data available for pharyngeal and rectal infections. Although Zoliflodacin has demonstrated therapeutic efficacy for gonococcal urogenital and rectal infections, further data on the treatment of pharyngeal gonorrhoea would be useful due to different pharmacokinetic and microbiological obstacles at this anatomical site1, 5.
SAFETY AND ADVERSE EFFECTS
Zoliflodacin safety profile has been well demonstrated in phase 1, 2, and 3 clinical trials, producing favourable tolerability for use. During phase 1 studies on single doses of 3 g and 4 g given with a high-fat meal, 42% of the group had 34 treatment-emergent adverse events (TEAEs), with all classified as mild.17 Headache was identified as the most common adverse event, reported by 45.8% of subjects (22 out of 48), and mild other side effects were reported16.
The phase 3 trial comparing Zoliflodacin with ceftriaxone plus azithromycin validated the safety of the drug in the targeted patient population with uncomplicated urogenital gonorrhoea14,17. The trial showed that Zoliflodacin is well tolerated, with a safety profile similar to usual therapy17. There were no serious adverse incidents due to Zoliflodacin reported, and both the frequency and intensity of adverse events mirrored earlier phase studies.
The cardiac safety has further been assessed when a comprehensive QT study investigates the impact of Zoliflodacin on cardiac repolarization in healthy adult subjects18. Although the literature does not contain high-level results, the nature of this study was regulatory-required, and the subsequent phase 3 trials suggest no relevant clinical issues in QT prolongation.
Also, the beneficial safety profile applies to pharmacokinetic studies investigating drug-drug interactions. However, it was confirmed by population pharmacokinetics that the co-administration of strong CYP3A4 inhibitors was indeed a risk factor for exposure, but it was not a cause for concern for drug safety, as it did not require dose changes. The predominance of the faecal elimination pathway (79.6% of dose) and low renal excretion of unchanged drug (2.5%) indicates little risk of drug accumulation in patients with renal impairment, though specific studies in this population have not been extensively reported11.
In total, safety evidence indicated Zoliflodacin as a well-tolerated antibiotic for single-dose outpatient gonorrhoea treatment. The mild and transient nature of reported adverse events and the convenience of oral administration favours Zoliflodacin compared to intramuscular ceftriaxone, which may have injection site reactions and would require administration in healthcare facilities16,17.
RESISTANCE PATTERNS
Key to Zoliflodacin is the high conservation of its target, GyrB, and the few resistance mutations to date. Decades of genomic surveillance of 27,151 global gonococcal isolates between 1928 and 2021 showed that 97.0% had wild-type GyrB sequences(19). Amino acid modifications of GyrB were infrequent, and only one isolate (0.0037%) contained a replacement at a resistance-associated codon (D429V); this resulted in a Zoliflodacin MIC of 8 mg/L19. None of the other detected mutations led to MIC values outside the wild-type distribution, corroborating extremely high susceptibility over the course of nearly 100 years of gonococcal evolution19.
In vitro selection for drug resistance studies has revealed targeted GyrB mutations associated with decreased Zoliflodacin sensitivity. The three most frequently encountered resistance mutations are D429N, K450T, and K450N, and they confer MICs of 0.5-4 mg/L for Zoliflodacin(20). Zoliflodacin-resistance mutations occur with low prevalence when a single agent is used, and fewer mutations are observed during combination therapy20. Significantly, resistant mutants chosen in vitro demonstrate substantially lower biological fitness than susceptible parent strains, indicating that resistant strains may not perform as well in vivo12.
S467N substitution in GyrB is a naturally occurring polymorphism that confers reduced susceptibility without conferring frank resistance. Strains harbouring this substitution (MIC = 0.25 mg/L) are prone to additional resistance mutations, especially D429N, after they have been exposed to suboptimal Zoliflodacin doses12. Pharmacodynamic investigations showed that the strains with S467N need a dose ≥ 3 g of Zoliflodacin for their effective eradication12. Of the 986 China isolates tested between 2014 and 2018, a single isolate with the S467N mutation was identified, and no D429N/A or K450T mutations were detected.6
Cross-resistance profiles are especially favourable for Zoliflodacin. Despite both Zoliflodacin and fluoroquinolones targeting type II topoisomerases, no cross-resistance has been observed between these drug classes.5,10 Fluoroquinolone resistance mutations in GyrA and ParC do not notably affect Zoliflodacin MICs5. Mutations that lead to the overexpression of the MtrCDE efflux pump, conferring resistance to several antibiotic classes, do not significantly impact Zoliflodacin susceptibility5. This lack of cross-resistance is important, as it allows Zoliflodacin to preserve its full activity against multi-drug-resistant strains of gonococcal pathogens that have exhausted other available treatment strategies.6,9,10
All the above-described antimicrobial resistance targets, key mechanisms and cross resistance were enlisted in Table-3.
Zoliflodacin and Gepotidacin, new topoisomerase inhibitors being developed for gonorrhoea, are both now under investigation for possible cross-resistance21. Both products target bacterial topoisomerases, but the two drugs have distinct binding sites and mechanisms that limit cross-resistance potential, which is still under investigation21.
Table 3 : Antimicrobials showing resistance mechanisms and cross resistance patterns
|
Antibiotic Class |
Primary target |
Key Resistance Mechanisms |
Zoliflodacin Cross-Resistance |
Prevalence of Resistance |
|
Zoliflodacin |
GyrB (DNA gyrase) |
GyrB mutations (D429N, R450H) |
— |
<0.1% |
|
Fluoroquinolones |
GyrA, ParC |
GyrA/ParC mutations (S91, D95) |
No |
49.9% |
|
Cephalosporins |
PBP2 (penA) |
Mosaic penA alleles |
No |
0.2–1.6% |
|
Macrolides |
23S rRNA |
23S rRNA mutations |
No |
6.7% |
|
Tetracyclines |
16S rRNA |
tetM determinant |
No |
15–20% |
|
Aminoglycosides |
16S rRNA |
Enzymatic modification |
No |
<5% |
|
Gepotidacin |
GyrA, ParE |
GyrA/ParE mutations |
Partial* |
<1% |
*Some laboratory-selected mutants show partial cross-resistance between Zoliflodacin and Gepotidacin.GyrB = DNA gyrase subunit B; ParC/ParE = Topoisomerase IV subunits; PBP = Penicillin-binding protein
In-depth surveillance of multiple geographic regions shows evidence for sustained susceptibility over time, but slight temporal variations demand monitoring. In China, while the percentage of isolates with higher Zoliflodacin MICs increased from 2014 to 2018, all isolates remained within the previously described wild-type MIC distribution6. This trend emphasizes the necessity for ongoing surveillance and antimicrobial stewardship to maintain the effectiveness of Zoliflodacin17.
Comparative Studies with Other Antibiotics: Comparative susceptibility testing in vitro has further substantiated the activity of Zoliflodacin in comparison to other antimicrobials.
Table 4: Comparative Antimicrobial activity against N. gonorrhoea
|
Antibiotic |
Class |
MIC₅₀ (µg/mL) |
MIC₉₀ (µg/mL) |
Resistance Rate (%) |
Route |
Status |
|
Zolifloda |
Spiropyrimidinetrione |
0.06 |
0.125 |
<0.1 |
Oral |
Phase 3 complete |
|
Ceftriaxo |
Cephalosporin |
0.004 |
0.008 |
0.2 |
IM |
Current standard |
|
Cefixime |
Cephalosporin |
0.03 |
0.125 |
1.6 |
Oral |
Not recommended |
|
Azithromycin |
Macrolide |
0.5 |
4.0 |
6.7 |
Oral |
Combination only |
|
Ciprofloxacin |
Fluoroquinolone |
8.0 |
32.0 |
49.9 |
Oral |
Not recommended |
|
Gentamicin |
Aminoglycoside |
4.0 |
8.0 |
<5 |
IM |
Alternative |
|
Gepotidacin |
Triazaacenaphthylene |
0.5 |
1.0 |
<1 |
Oral |
Phase 3 |
|
Solithromycin |
Fluoroketolide |
0.25 |
0.5 |
<1 |
Oral |
Development |
MIC values and resistance rates based on recent surveillance data (2018-2024), IM = Intramuscular.
The table.4 summarizes the resistance rate and MIC50 & MIC90 of various antimicrobials. In vitro comparisons demonstrate that while several current therapies face significant resistance thresholds, Zoliflodacin remains highly potent with no evidence of cross-resistance to existing antibiotic classes.
The absence of cross-resistance with pre-existing antimicrobials is an important comparative advantage. Fluoroquinolones, macrolides, and extended-spectrum cephalosporins have also developed progressive resistance; yet, Zoliflodacin demonstrates a unique pathway with the target preserved, allowing it to retain activity despite the emergence of class resistance5,9. This is especially true of the extensively drug-resistant strains, where Zoliflodacin remains totally active6,15.
Studies that assess the compatibility of Zoliflodacin with other antimicrobials showed that in vitro testing of Zoliflodacin combined with six therapeutically relevant antimicrobials demonstrated that the majority of the drug combinations rapidly inhibited N. gonorrhoeae growth20. Nonetheless, the Zoliflodacin kill rate of static in vitro assays was lower with tetracycline or azithromycin combined with Zoliflodacin20. Crucially, dynamic hollow fibre infection model studies, which more accurately simulate in vivo conditions, demonstrated that Zoliflodacin plus doxycycline combination therapy was slightly more effective than monotherapy and suppressed the emergence of resistance15. This is clinically relevant considering the frequent co-administration of doxycycline for chlamydial coinfection15
Comparing side effects to the LOS drug in clinical practice, the pharmacokinetic advantages are oral bioavailability, single-dose treatment, and predictable dose-proportional exposure11. Zoliflodacin does not require intramuscular or institution-provided intramuscular injection and is self-administered orally, as opposed to ceftriaxone, increasing access and patient acceptance.17 Its pharmacokinetic profile supports a high probability of target attainment across the MIC distribution of clinical isolates, providing confidence in consistent efficacy.
Drug Development and Regulatory Status:
Zoliflodacin is developed and promoted in concert with global health partners in the pharmaceutical industry. The development of the drug was established in cooperation with the Global Antibiotic Research and Development Partnership (GARDP), reflecting the public health imperative to develop new treatments for antibiotic-resistant infections.7,17 This partnership model has been instrumental in progressing antibiotics against priority pathogens for which commercial promotion alone sometimes may not provide the necessary support17.
Phase 1, 2 and 3 studies as clinical development program has been a stepwise process. The initial phase 1 study determined safety, tolerability, and pharmacokinetic parameters in healthy volunteers11. The phase 2 proof-of-concept trials showed effectiveness for single-dose therapy of urogenital gonorrhoeae and provided reliable evidence for dose selection for crucial trials13. The phase 3 program was an international, randomized, controlled trial that demonstrated non-inferiority to standard therapy14.
Zoliflodacin got it’s approval from FDA on December 12, 2025, with an official brand name of “Nuzolvence” as a first-in-class Spiropyrimidinetrione going to be used in uncomplicated gonorrhoea, as of from the most recent reporting. Further, in 2026, phase-3 clinical trial results were published showing that Zoliflodacin was not inferior to ceftriaxone plus azithromycin14. With this milestone, a major step towards regulatory approval by USFDA was made. This drug is poised to become one of the first entirely novel chemical entities to be approved against gram-negative bacteria in the 21st century4.
Expert advice on the optimal introduction of Zoliflodacin has identified that implementation strategies need to be extensive17. The forthcoming World Health Organization guidelines will likely provide recommendations for the use of Zoliflodacin in clinical practice.17
Safety assessments, along with QT studies to confirm cardiac safety, and a well-characterized pharmacokinetics to inform dosing recommendations, have all been incorporated into the regulatory pathway. Population pharmacokinetic studies from data of various trials yielded strong support towards single-dose regimen of 3 g.18
Intensive surveillance of gonococcal susceptibility and resistance trajectories has driven the development of zoliflodacin. Besides in vitro assays, in silico mining of over 27,000 gonococcal genomes confirmed high conservation of GyrB targets and low occurrence of resistance mutations. This genomic monitoring conveys confidence in the probable durability of Zoliflodacin and provides direction for resistance surveillance post-approval.19
The position of the drug within the overall field of gonorrhoea care underscores not only the clinical benefits, but also the immediate need for alternate therapeutic options. Zoliflodacin therefore fills an important gap in the antimicrobial community, with N. gonorrhoeae identified as one of the targets for WHO priorities and as a member of the “WHO Priority Pathogens List for Research and Development of New Antibiotics”.2
FUTURE PERSPECTIVES AND CONCLUSIONS
Due to its novel mode of action, strong activity against multidrug-resistant strains, and a favourable safety profile, zoliflodacin is an important advancement in the treatment of gonorrhoea. The successful completion of phase 3 trials demonstrating non-inferiority to the existing standard treatment represents a major development in addressing the global threat of antibiotic-resistant N. gonorrhoeae.14,17
Several elements enable favourable indications for clinical use with zoliflodacin. Oral bioavailability and a single-dose regimen of the drug offer significant practical advantages over intramuscular ceftriaxone that may potentially contribute to increased treatment accessibility, patient acceptability, and adherence17. Due to the lack of cross-resistance with existing antimicrobials, zoliflodacin is fully active against strains resistant to fluoroquinolones, macrolides, and cephalosporins5,9. The high conservation of the GyrB target and low prevalence of resistance mutations observed across nearly a century of gonococcal evolution indicate the possibility for prolonged efficacy19.
But there are some areas for continued research and follow-up. More information on the treatment of pharyngeal gonorrhoea would be needed as this site has unique pharmacokinetic and microbiological challenges5. Testing the susceptibility of Zoliflodacin in long-term studies should be required to detect any resistance and to inform stewardship strategies6,17. Not yet clinically relevant, but the modest trend of higher MICs over time reported in some surveillance studies highlights the importance of continued monitoring6.
The drug’s potential as a combination therapy is an important domain to investigate as the pipeline advances. An experimental study of the Zoliflodacin-doxycycline combination treatment in a dynamic hollow fibre infection model was used to demonstrate its efficacy and suggested that resistance could be limited15. Due to high rates of gonorrhoea-chlamydia co-infection, this protocol may exert practical implications in the clinic15. Other clinical studies on combination regimens would be useful.
Other potential uses of Zoliflodacin beyond gonorrhoea should be explored further. Initial results demonstrating therapeutic activity against Helicobacter pylori and Acinetobacter baumannii suggest applicability to other bacterial infections14,22. While clinical development is appropriately tailored to address the urgent need for new gonorrhoea therapeutics, the Spiropyrimidinetrione scaffold can serve as the basis for the development of agents targeting other gram-negative pathogens4.
Zoliflodacin formulation also provides important lessons for the overall development of antibiotics. Characterization of mechanisms, resistance patterns, and pharmacokinetics also establish some strong scaffolding to support stewardship and prevent resistance prior to widespread clinical application12,19. Zoliflodacin will need to come in an optimal method across multiple domains. Improved surveillance capabilities in place for susceptibility monitoring and resistance emergence have been proposed17,19.
Based on this data it is expected that this is a significant breakthrough in the battle against antibiotic resistance in gonorrhoea globally. Along with its unique mechanism, potent activity, and safety profile, the drug will broaden therapeutic options with practical benefits. Effective clinical use would require cautious stewardship, comprehensive surveillance, ongoing exploration, and further evaluation to optimize its use while maintaining its utility and efficacy. As one of the very first novel antibiotics targeting gram-negative bacteria to attain late-stage development in several decades, Zoliflodacin is promising for patients and clinicians who face untreatable gonorrhoea and suggests potential clinical utility in the future2,4.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST STATEMENT:
Authors declare no conflict of interest.
SOURCE OF FUNDING:
None
AUTHORS’ CONTRIBUTIONS:
All authors (BSN, MC, GR, AM, KS, YR, VR, AP) contributed substantially to the conception and design of the work, data acquisition and interpretation, manuscript drafting and critical revision, approved the final version, and agree to be accountable for all aspects of the work.
DECLARATION FOR THE USE OF GENERATIVE ARTIFICIAL INTELLIGENCE (AI) IN SCIENTIFIC WRITING: Generative AI (Gemini) was used only for creating illustrative figures in this manuscript and reviewed by the authors for scientific accuracy.
REFERENCES
-
D. S, Verma A, Desai S. Zoliflodacin: a hope to treat antibiotic-resistant Neisseria gonorrhoeae. Infect Dis Res. 2022;3(2):10. doi:10.53388/IDR20220525010
-
Raccagni AR, Ranzenigo M, Bruzzesi E, Maci C, Castagna A, Nozza S. Neisseria gonorrhoeae Antimicrobial Resistance: The Future of Antibiotic Therapy. J Clin Med. 2023 Dec 18;12(24):7767. doi:10.3390/jcm12247767
-
Bradford PA, Miller AA, O’Donnell J, Mueller JP. Zoliflodacin: An Oral Spiropyrimidinetrione Antibiotic for the Treatment of Neisseria gonorrheae , Including Multi-Drug-Resistant Isolates. ACS Infect Dis. 2020 Jun 12;6(6):1332–45. doi:10.1021/acsinfecdis.0c00021
-
Morgan H, Lipka-Lloyd M, Warren AJ, Hughes N, Holmes J, Burton NP, et al. A 2.8 Å structure of zoliflodacin in a DNA-cleavage complex with Staphylococcus aureus DNA gyrase.
-
Unemo M, Ahlstrand J, Sánchez-Busó L, Day M, Aanensen D, Golparian D, et al. High susceptibility to zoliflodacin and conserved target (GyrB) for zoliflodacin among 1209 consecutive clinical Neisseria gonorrhoeae isolates from 25 European countries, 2018. J Antimicrob Chemother. 2021 Apr 13;76(5):1221–8. doi:10.1093/jac/dkab024
-
Le W, Su X, Lou X, Li X, Gong X, Wang B, et al. Susceptibility Trends of Zoliflodacin against Multidrug-Resistant Neisseria gonorrhoeae Clinical Isolates in Nanjing, China, 2014 to 2018. Antimicrob Agents Chemother. 2021 Feb 17;65(3):e00863-20. doi:10.1128/AAC.00863-20
-
Thakare, R., Dasgupta, A., Chopra, S. Zoliflodacin. DNA gyrase subunit B (GyrB) (bacterial) inhibitor, Treatment of uncomplicated gonorrhea. Drugs Future. 2019;44(12):943. doi:10.1358/dof.2019.44.12.3051831
-
Collins JA, Basarab GS, Chibale K, Osheroff N. Interactions between Zoliflodacin and Neisseria gonorrhoeae Gyrase and Topoisomerase IV: Enzymological Basis for Cellular Targeting. ACS Infect Dis. 2024 Aug 9;10(8):3071–82. doi:10.1021/acsinfecdis.4c00438
-
Matthew F, Stella MM, Tandarto K, Regina R. Zoliflodacin, A Novel Antimirobial Agent Against neisseria Gonorhoeae: A Systematic Rreview of Quasi-Experimental Studies. Berk Ilmu Kesehat Kulit Dan Kelamin. 2024 Mar 31;36(1):68–74. doi:10.20473/bikk.V36.1.2024.68-74
-
Roh KH, Luong ND, Liu C, Seo YH, Lee H, Unemo M, et al. In-vitro Activities of Zoliflodacin and Solithromycin Against Neisseria gonorrhoeae Isolates from Korea. Ann Lab Med. 2025 Nov 1;45(6):626–9. doi:10.3343/alm.2024.0522
-
O’Donnell J, Lawrence K, Vishwanathan K, Hosagrahara V, Mueller JP. Single-Dose Pharmacokinetics, Excretion, and Metabolism of Zoliflodacin, a Novel Spiropyrimidinetrione Antibiotic, in Healthy Volunteers. Antimicrob Agents Chemother. 2019 Jan;63(1):e01808-18. doi:10.1128/AAC.01808-18
-
Jacobsson S, Golparian D, Oxelbark J, Franceschi F, Brown D, Louie A, et al. Pharmacodynamic Evaluation of Zoliflodacin Treatment of Neisseria gonorrhoeae Strains With Amino Acid Substitutions in the Zoliflodacin Target GyrB Using a Dynamic Hollow Fiber Infection Model. Front Pharmacol. 2022 Apr 14;13:874176. doi:10.3389/fphar.2022.874176
-
Taylor SN, Marrazzo J, Batteiger BE, Hook EW, Seña AC, Long J, et al. Single-Dose Zoliflodacin (ETX0914) for Treatment of Urogenital Gonorrhea. N Engl J Med. 2018 Nov 8;379(19):1835–45. doi:10.1056/NEJMoa1706988
-
Luckey A, Balasegaram M, Barbee LA, Batteiger TA, Broadhurst H, Cohen SE, et al. Zoliflodacin versus ceftriaxone plus azithromycin for treatment of uncomplicated urogenital gonorrhoea: an international, randomised, controlled, open-label, phase 3, non-inferiority clinical trial. The Lancet. 2026 Jan;407(10524):147–60. doi:10.1016/S0140-6736(25)01953-1
-
Jacobsson S, Golparian D, Oxelbark J, Kong FYS, Da Costa RMA, Franceschi F, et al. Pharmacodynamics of zoliflodacin plus doxycycline combination therapy against Neisseria gonorrhoeae in a gonococcal hollow-fiber infection model. Front Pharmacol. 2023 Dec 7;14:1291885. doi:10.3389/fphar.2023.1291885
-
Luckey A, Alirol E, Delhomme S, O’Donnell J, Bettiol E, Mueller J, et al. Effect of food on the pharmacokinetics of zoliflodacin granules for oral suspension: Phase I open‐label randomized cross‐over study in healthy subjects. Clin Transl Sci. 2023 May;16(5):770–80. doi:10.1111/cts.13487
-
Pascual F, Au C, Chikwari CD, Daram P, Deal C, Miranda AE, et al. Recommendations for the optimal introduction of novel antibiotics to treat uncomplicated gonorrhoea in the face of increasing antimicrobial resistance: a case study with zoliflodacin. BMC Glob Public Health. 2024 Sep 3;2(1):58. doi:10.1186/s44263-024-00087-w
-
Newman LM, Kankam M, Nakamura A, Conrad T, Mueller J, O’Donnell J, et al. Thorough QT Study To Evaluate the Effect of Zoliflodacin, a Novel Therapeutic for Gonorrhea, on Cardiac Repolarization in Healthy Adults. Antimicrob Agents Chemother. 2021 Nov 17;65(12):e01292-21. doi:10.1128/AAC.01292-21
-
Golparian D, Jacobsson S, Sánchez-Busó L, Bazzo ML, Lan PT, Galarza P, et al. GyrB in silico mining in 27 151 global gonococcal genomes from 1928–2021 combined with zoliflodacin in vitro testing of 71 international gonococcal isolates with different GyrB, ParC and ParE substitutions confirms high susceptibility. J Antimicrob Chemother. 2023 Jan 5;78(1):150–4. doi:10.1093/jac/dkac366
-
Foerster S, Drusano G, Golparian D, Neely M, Piddock LJV, Alirol E, et al. In vitro antimicrobial combination testing of and evolution of resistance to the first-in-class spiropyrimidinetrione zoliflodacin combined with six therapeutically relevant antimicrobials for Neisseria gonorrhoeae. J Antimicrob Chemother. 2019 Dec 1;74(12):3521–9. doi:10.1093/jac/dkz376
-
Mukherjee A, Blomqvist SO, Helekal D, Das AA, Palace SG, Grad YH. Zoliflodacin and gepotidacin cross-resistance in Neisseria gonorrhoeae. 2025 Nov 25. doi:10.1101/2025.11.25.690462
-
Liu J, Jia J, Shi T, Bai Y, Huang Y, Zeng L, et al. Evaluating the potency of zoliflodacin against Helicobacter pylori : In vitro activity and conserved GyrB target. Helicobacter. 2024 Mar;29(2):e13075. doi:10.1111/hel.13075
Submit a Manuscript:
Copyright © Author(s) 2026. JASPI- Journal of Antimicrobial Stewardship Practices and Infectious Diseases.

