Profile and Assessment of Potential Risk Factors of Neonatal Candidemia Cases in a Tertiary-care Hospital in North India
JASPI March 2026 / Volume 4 /Issue 1
Copyright: © Author(s). This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Firoze S, Khan F, Ahmed S, et al. Profile and Assessment of Potential Risk Factors of Neonatal Candidemia Cases in a Tertiary-care Hospital in North India. JASPI. 2026;4(1):Page No DOI: 10.62541/jaspi108
ABSTRACT
Introduction: Candida blood stream infections (BSIs) are a major contributing factor to neonatal sepsis and sepsis-related morbidities. Despite advances in newborn care, there remains a considerable morbidity, a trend towards neonatal candidemia, and occasional cases of antifungal resistance.
Aim: Our study aimed to identify the prevalent Candida species, antifungal susceptibility (AFS) patterns, and to ascertain risk factors associated with candida BSIs in neonates.
Materials and Methods: This was an eighteen-month retrospective study of candidemia in neonates. Positive aerobic BactT-Alert blood cultures having yeast were speciated (HiMedia Chrom-Agar and Vitek2®). AFS of isolates was performed using VITEK2® and disc diffusion.
Results: 303 (11.1%) out of 2748 positive blood cultures revealed Candida species, from which 84 (27.7%) were confirmed as newborn (0 to 28 days) BSI cases. Candida tropicalis (29.8%) was the most common isolate, followed by Candida albicans (26.2%) and C. glabrata (25%). AFS of these isolates revealed overall resistance of amphotericin B as 2.4% and of fluconazole as 13.1%. 60 isolates tested by Vitek 2 were sensitive to voriconazole; there was 5% caspofungin, 1.7% micafungin, and 1.7% flucytosine resistance. Risk factor assessment included NICU admission (60.7%), broad-spectrum antibiotic usage (71.4%), low birth weight (65.5%), prematurity (65.5%,), ventilator support (44%) and mortality (23.8%).
Conclusion: The rise in resistance and neonatal candidemia emphasises the necessity of reevaluating the application of stringent infection control methods, suitable prophylactic antifungals, and a limited antibiotic-use strategy, among other preventative, as well as therapeutic approaches.
KEYWORDS: Candidemia; Neonates; Antifungal; Resistance; Risk factors
INTRODUCTION
Newborn survival has significantly increased as a result of advancements in neonatal care. Nonetheless, both bacterial and mycological systemic infections with early (within 72 hours) and late (post 72 hours) onset still pose a serious risk and are a major contributor to neonatal morbidity1. There is growing recognition of the role of Candida species in neonatal intensive care units (NICUs). It is responsible for 9 to 13% of neonatal blood-stream infections (BSI) and is the third most frequent cause of late-onset sepsis in NICU patients2.
Although Candida albicans has traditionally been the most frequently isolated species, non-albicans Candida (NAC), including C. glabrata, C. krusei, C. parapsilosis, and C. tropicalis, have recently emerged as important opportunistic pathogens3. This shift in epidemiology is concerning, as NAC resistance to azoles is increasingly recognized and appears to be linked to the widespread use of azole agents4. Moreover, several NAC species demonstrate cross-resistance to newer triazoles, while others exhibit intrinsic resistance to fluconazole (FLZ). Hence, it is essential that all yeast isolates, whether from blood or other clinical specimens, undergo both antifungal susceptibility testing and species-level identification 3,5.
In neonatal intensive care units (NICUs), candidiasis is believed to arise primarily from endogenous colonization, given the high prevalence of fungal acquisition in this population. Within the first week of life, approximately 10% of neonates become colonized, a figure that may rise to nearly 64% after a four-week hospital stay6,7. Outbreaks in NICUs have also been attributed to contaminated injectable preparations, such as lipid emulsions used for total parenteral nutrition, or via transmission from colonized healthcare personnel and patient-to-patient spread, particularly in the context of inadequate hand hygiene8-10. Other recognized risk factors include mechanical ventilation, broad-spectrum antibiotic therapy, prior gastrointestinal surgery, indwelling devices, low birth weight (LBW), and prematurity11,12.
Clinically, Candida bloodstream infection often resembles bacterial sepsis, making diagnosis challenging. Mycotic sepsis may present with nonspecific features such as lethargy, glycemic instability, neutropenia, and respiratory distress. Disseminated fungal disease can result in extensive end-organ involvement, affecting the kidneys, liver, lungs, brain, eyes, bones, spleen, and joints. Notably, invasive infection may occur even in the absence of positive culture results13. Renal involvement may manifest as acute dysfunction, hypertension, or flank masses, while ocular dissemination can present as endophthalmitis, a severe complication requiring immediate medical intervention14,15.
The crude death rate linked to candidemia is around 60%, whereas the attributable mortality rate may reach 49%. The at-risk group involved, the quality of medical care, and the resistance or susceptibilities of different Candida species and their strains to certain antifungal drugs are some of the variables that can affect the incidence and related mortality of Candida BSI16. Owing to significant regional variations, local epidemiology expertise is essential for managing and preventing invasive infections caused by Candida17.
This study aimed to identify the various Candida pathogens, examine their sensitivity patterns, and assess risk factors associated with neonatal candidemi
MATERIALS AND METHODS
Study Design and Setting: This (January 2023 to June 2024), retrospective-observational research was conducted at the Microbiology unit of a tertiary-care hospital located in a city of northern India. The study was approved by the Institutional Ethics Committee of Jawaharlal Nehru Medical College, Aligarh, India (IEC Approval No: IECJNMC/1078 ). Candida species were discovered in 303 (11.1%) of the 2748 blood culture bottles that were labelled positive during the study period (Figure 1). Of these, 90 were isolated from neonate’s blood samples that ranged in age from 0 to 28 days. 6 samples were excluded because they lacked corroborated clinical characteristics or because they revealed yeast as a contamination or commensal. This research took into account 84 (27.7%) neonates who showed signs and symptoms of infection such as lethargy, feed intolerance, failure to thrive or sepsis combined with at least one positive blood culture for Candida species.
Laboratory Procedures: Blood specimens were obtained for culture, while adhering to aseptic procedures after obtaining informed consents from the guardians of the patients. Automated BacTAlert3D (Biomerieux, France) was used to perform the blood culture. A paired blood culture was taken. Upon flagging of the blood culture bottle, direct microscopy was done using Gram’s stain followed by inoculation onto 5% sheep blood agar incubated at 37°C, along with two plates of Sabouraud dextrose agar incubated at 25°C and 37°C. Growth of colonies was visible in 24 hours for all cases. A number of phenotypic assays were run to determine the isolates of Candida species. These comprised of CHROMagar Candida (CHROMagar, Paris, France), chlamydospore formation on cornmeal agar (HiMedia, India), Gram’s staining, and germ tube visualization.
Antifungal Susceptibility Testing: The Clinical and Laboratory Standards Institute (CLSI) standards18 were followed in the disk diffusion method (Voriconazole – 1µg, Fluconazole – 25µg, Amphotericin B – 10µg, Caspofungin – 5µg, Itraconazole – 10 µg) used to determine the antifungal susceptibility of 24 isolates. Because the hospital serves a highly impoverished community, financial constraints prevented Vitek from being used on all samples. Vitek 2 Compact (Biomerieux, France) was used to identify 60 samples through the use of ID-YST and YS08 (Voriconazole – 0.12-8 µg/mL, Fluconazole – 1-64 µg/mL, Amphotericin B – 0.25-16 µg/mL and 5-Flucytosine – 1-64 µg/mL, Caspofungin – 0.125 -8 µg/mL, Micafungin – 0.06-8 µg/mL) cards for AFS testing.
Statistics: Statistical analysis was done using Statistical Package for Social Sciences (SPSS) version 20 and the prevalence of organisms was determined and expressed in percentage. Categorical variables were summarized with frequency and continuous variables were summarized with mean and standard deviation. For categorical outcomes, comparison between groups were performed using the Chi-square test or Fisher’s exact test, as appropriate. A p-value of <0.05 was considered statistically significant.
OBSERVATIONS AND RESULTS
Neonates accounted for 90 of the 303 Candida isolates, of which 84 (27.7%) were proven to originate from cases of neonatal bloodstream infection. Likewise, among the 2445 aerobic bacterial isolates, 241 were from neonates, with 234 (9.6%) confirmed as being from neonate BSI cases (Figure 1 & Table 1).
Figure 1. Flagged Blood Culture Bottles and Candida in Blood Cultures from Neonates (0 to 28 days)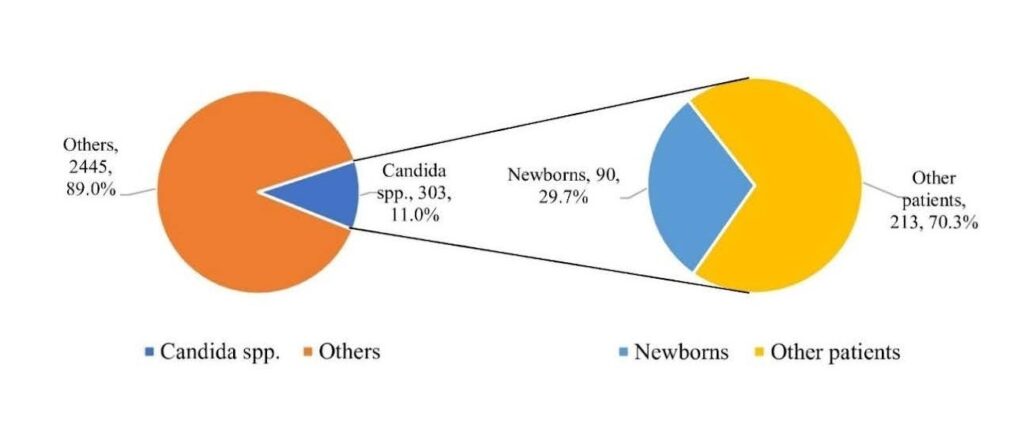
Table 1. Distribution of pure isolates from neonatal septicemia cases (n = 318)
Organism | No. of isolates | Percentage | |
Candida species (n = 84) | Candida albicans | 22 | 6.9% |
C. glabrata | 21 | 6.6% | |
C. krusei | 4 | 1.3% | |
C. parapsilosis | 9 | 2.8% | |
C. pelliculosa | 3 | 0.9% | |
C. tropicalis | 25 | 7.9% | |
Bacterial isolates (n = 234) | Enterobacteriaceae (n = 84) | ||
Citrobacter sp. | 6 | 1.9% | |
Escherichia coli | 14 | 4.4% | |
Enterobacter sp. | 2 | 0.6% | |
Klebsiella | 59 | 18.6% | |
Serratia sp. | 3 | 0.9% | |
Gram positive cocci (n = 106) | |||
Enterococci | 19 | 6% | |
Other Staphylococcus sp. | 62 | 19.5% | |
Staphylococcus aureus | 24 | 7.5% | |
Streptococci | 1 | 0.3% | |
Nil-fermenters (n = 44) | |||
Acinetobacter | 21 | 6.6% | |
Pseudomonas | 21 | 6.6% | |
Stenotrophomonas sp. | 2 | 0.6% | |
Organism | No. of isolates | Percentage | |
Candida species (n = 84) | Candida albicans | 22 | 6.9% |
C. glabrata | 21 | 6.6% | |
C. krusei | 4 | 1.3% | |
C. parapsilosis | 9 | 2.8% | |
C. pelliculosa | 3 | 0.9% | |
C. tropicalis | 25 | 7.9% | |
Bacterial isolates (n = 234) | Enterobacteriaceae (n = 84) | ||
Citrobacter sp. | 6 | 1.9% | |
Escherichia coli | 14 | 4.4% | |
Enterobacter sp. | 2 | 0.6% | |
Klebsiella | 59 | 18.6% | |
Serratia sp. | 3 | 0.9% | |
Gram positive cocci (n = 106) | |||
Enterococci | 19 | 6% | |
Other Staphylococcus sp. | 62 | 19.5% | |
Staphylococcus aureus | 24 | 7.5% | |
Streptococci | 1 | 0.3% | |
Nil-fermenters (n = 44) | |||
Acinetobacter | 21 | 6.6% | |
Pseudomonas | 21 | 6.6% | |
Stenotrophomonas sp. | 2 | 0.6% | |
The most common pathogens isolated were other Staphylococcus species (n= 62, 19.5%), Klebsiella species (n= 59, 18.6%), Candida tropicalis (n= 25, 7.9%), Staphylococcus aureus (n= 24, 7.5%), C. albicans (n= 22, 6.9%), and C. glabrata, Acinetobacter species, and Pseudomonas species (n= 21 each, 6.6%).
Non-albicans Candida species were responsible for 73.8% of the 84 newborn Candida BSI cases. C. tropicalis accounted for 25 (29.8%) of the total species (Table 2).
Table 2. Distribution of Candida species isolated from blood of neonates (n = 84)
Organism | No of isolates | Percentage | ||
Candida albicans | 22 | 26.2% | ||
Non- (NAC) | C. glabrata | 21 | 25% | Total NAC = 73.8% |
C. krusei | 4 | 4.8% | ||
C. parapsilosis | 9 | 10.7% | ||
C. pelliculosa | 3 | 3.6% | ||
C. tropicalis | 25 | 29.8% | ||
The distribution of the other isolated species was: Candida albicans, 22 (26.2%); C. glabrata, 21 (25%); C. parapsilosis, 9 (10.7%); C. krusei, 4 (4.8%); and Candida pelliculosa, 3 (3.6%).
Table 3. Combined Antifungal Susceptibility Profile of Candida.
Organism | FLZ (n=84) | AMB (n=84) | VOR n=(60) | CSF n=(60) | MYC n=(60) | 5FC n=(60) | ||||||
S | R | S | R | S | R | S | R | S | R | S | R | |
C. albicans (22) | 20/22 (90.9%) | 2/19 (9.1%) | 22/22 (100%) | 0 (0) | 19/19 (100%) | 0 (0) | 18/19 (94.7%) | 1/19 (5.3%) | 18/19 (94.7%) | 1/19 (5.3%) | 18/19 (94.7%) | 1/19 (5.3%) |
C. glabrata (21) | 21/21 (100%) | 0 (0) | 21/21 (100%) | 0 (0) | 18/18 (100%) | 0 (0) | 18/18 (100%) | 0 (0) | 18/18 (100%) | 0 (0) | 18/18 (100%) | 0 (0) |
C. krusei 4) | 0/4 (0) | 4/4 (100%) | 3/4 (75%) | 1/4 (25%) | 2/2 (100%) | 0 (0) | 2/2 (100%) | 0 (0) | 2/2 (100%) | 0 (0) | 2/2 (100%) | 0 (0) |
C. parapsilosis (9) | 8/9 (88.8%) | 1/9 (11.2%) | 9/9 (100%) | 0 (0) | 5/5 (100%) | 0 (0) | 5/5 (100%) | 0 (0) | 5/5 (100%) | 0 (0) | 5/5 (100%) | 0 (0) |
C. pelliculosa (3) | 3/3 (100%) | 0 (0) | 2/3 (66.7%) | 1/3 (33.3%) | 3/3 (100%) | 0 (0) | 3/3 (100%) | 0 (0) | 3/3 (100%) | 0 (0) | 3/3 (100%) | 0 (0) |
C. tropicalis (25) | 21/25 (84%) | 4/25 (16%) | 13/13 (100%) | 0 (0) | 13/13 (100%) | 0 (0) | 11/13 (84.6%) | 2/13 (15.4%) | 13/13 (100%) | 0 (0) | 13/13 (100%) | 0 (0) |
73 (86.9%) | 11 (13.1%) | 82 (97.6%) | 2 (2.4%) | 60 (100%) | 0 (0) | 57 (95%) | 3 (5%) | 59 (98.3%) | 1 (1.7%) | 59 (98.3%) | 1 (1.7%) | |
Among the 24 Candida isolates that were tested for antifungal susceptibility by disk diffusion method (n= 24), resistance to fluconazole, amphotericin B, was 20.8% and 0%, respectively. All of the C. albicans isolates were sensitive to fluconazole and amphotericin B. No resistance was seen in C. glabrata, to fluconazole and amphotericin B. All of the 2 isolates of C. krusei were resistant to fluconazole (100%); no resistance was seen with amphotericin B. All the C. parapsilosis isolates were sensitive to all antifungals, except for 1 isolate showing FLZ resistance (25%). Out of 12 C. tropicalis isolates tested, resistance was seen with FLZ (2; 16.7%).
Among the 60 Candida isolates that were tested for antifungal susceptibility by Vitek 2 (Table 3), (n = 60), resistance to fluconazole (FLZ), amphotericin B (AMB), voriconazole (VOR), caspofungin (CSF), micafungin (MYC), and flucytosine (5FC), was 10%, 3.3%, 0%, 5%, 1.7%, and 1.7%, respectively. All of the C. albicans isolates were sensitive to amphotericin B and voriconazole. 2 (10.5%) isolates were resistant to FLZ, while 1 (5.3%) isolate of C. albicans was resistant to CSF, MYC, 5FC, each. All the isolates of Candida glabrata and Candida parapsilosis were sensitive to FLZ, AMB, VOR, CSF, MYC, and 5FC. Both isolates of C. krusei were resistant to fluconazole, and 1 (50%) was resistant to amphotericin B. No resistance was seen with VOR, CSF, MYC, and 5FC. Only 1 (33.3%) isolate of C. pelliculosa was resistant to amphotericin B, while 2 (15.4%) isolates of C. tropicalis were resistant to fluconazole and caspofungin, each. All 3 C. pelliculosa isolates were sensitive to FLZ, VOR, CSF, MYC, and 5FC. All C. tropicalis isolates were sensitive to AMB, VOR, MYC, and 5FC.
Table 4. Demographic Data and Potential Risk Factors in Neonates | ||||||||||
Candidemia (n = 84) | Bacteraemia (n = 234) | P Value | ||||||||
No. | % | No. | % | |||||||
Admission | ||||||||||
NICU | 51 | 60.7% | 87 | 37.2% | <0.001 | |||||
Non-NICU | 33 | 39.3% | 147 | 62.8% | <0.001 | |||||
Antibiotics Usage | ||||||||||
< 7 days (d) | 18 | 60 | 21.4% | 71.4% | 27 | 150 | 11.5% | 64.1% | 0.013* | |
7 – 14 d | 32 | 38.1% | 40 | 17.1% | <0.001 | |||||
> 14 d | 10 | 11.9% | 83 | 35.5% | <0.001 | |||||
Not used | 24 | 28.6% | 84 | 35.9% | 0.112 | |||||
Birth Weight | ||||||||||
< 1 kg [ELBW] | 14 | 55 | 16.7% | 65.5% | 21 | 147 | 9% | 62.8% | 0.027* | |
1 – 1.5 kg [VLBW] | 16 | 19% | 47 | 20.1% | 0.419 | |||||
1.6 – 2.5 kg [LBW] | 25 | 29.8% | 79 | 33.8% | 0.251 | |||||
> 2.5 kg | 29 | 34.5% | 87 | 37.2% | 0.332 | |||||
Gender | ||||||||||
Males | 52 | 61.9% | 142 | 60.7% | 0.422 | |||||
Females | 32 | 38.1% | 92 | 39.3% | 0.422 | |||||
Gestational Age | ||||||||||
< 37 weeks (w) | 55 | 65.5% | 110 | 47% | 0.002* | |||||
37 w | 29 | 34.5% | 124 | 53% | 0.002* | |||||
Normal Vaginal Delivery | 72 | 85.7% | 210 | 89.7% | 0.159 | |||||
Outcome | ||||||||||
Alive | 64 | 76.2% | 207 | 88.5% | 0.003* | |||||
Death | 20 | 23.8% | 27 | 11.5% | 0.003* | |||||
Ventilator Support | ||||||||||
< 7 d | 7 | 37 | 8.3% | 44% | 57 | 104 | 24.4% | 44.4% | 0.001* | |
7 -14 d | 19 | 22.6% | 31 | 13.2% | 0.021* | |||||
> 14 d | 11 | 13.1% | 16 | 6.8% | 0.039* | |||||
Not used | 47 | 56% | 130 | 55.6% | 0.475 | |||||
ELBW= extremely low birth weight; VLBW= very low birth weight; LBW= low birth weight [* indicates P value <0.05]
A total of 318 neonates with bloodstream infection were analyzed, including 84 with candidemia and 234 with bacteremia (Table 4). NICU admission was significantly higher among neonates with candidemia compared to those with bacteremia (60.7% vs. 37.2%, p<0.001). Preterm birth (<37 weeks) was more frequent in the candidemia group (65.5% vs. 47%, p=0.002), with a significantly greater proportion of extremely low birth weight infants (<1 kg) (16.7% vs. 9%, p=0.027). Shorter prior antibiotic exposure (<7 days) was more common in candidemia (p=0.013), whereas prolonged mechanical ventilation (>7 days) was significantly associated with candidemia (p=0.021). Mortality was significantly higher in the candidemia cohort compared with bacteremia (23.8% vs. 11.5%, p=0.003). No statistically significant differences were observed with respect to gender, mode of delivery, or normal birth weight categories.
DISCUSSION
Pathogens isolated in our positive neonatal BSI samples included Staphylococcus species (19.5%), Klebsiella species (18.6%), Candida tropicalis (7.9%), Staphylococcus aureus (7.5%), Candida albicans (6.9%), Candida glabrata, Acinetobacter species, and Pseudomonas species (6.6% each). These findings closely parallel the observations of Nazir and Masoodi, who reported Staphylococcus species (16.6%), Klebsiella species (16.2%), Candida tropicalis (13.8%), Acinetobacter species (11.3%), Staphylococcus aureus (7.7%), and Candida albicans (5.6%) as the predominant isolates17. In the current research, 73.8% of the cases of newborn candidemia were caused by non-albicans Candida species, while 26.2% of cases were caused by C. albicans.
In our study, Candida tropicalis (29.8%) was the predominant species, followed by C. albicans (26.2%), C. glabrata (25%), C. parapsilosis (10.7%), C. krusei (4.8%), and C. pelliculosa (3.6%). Similar trends of C. tropicalis predominance have been reported nationally16, 19, 20, including by Nazir and Masoodi17 and Basu et al.21, whereas Juyal et al. observed C. parapsilosis as most common3. Contrastingly, Tunc et al. in Turkey reported C. albicans predominance30. The association of C. tropicalis with NICU outbreaks17, 23 underscores its virulence3. Fluconazole resistance (13.1%) was mainly among NAC, consistent with other Indian reports24-26, though all isolates remained 100% voriconazole susceptible19.
In contrast to our investigation, where fungal resistance to amphotericin B was found to be 2.4% across all Candida isolates, the Central India-based publication by Narain et al., showed that all fungal isolates were susceptible to the antifungals. Biswas et al., however, found comparable outcomes, citing a 3.6% resistance to amphotericin B19. Caspofungin, micafungin, and flucytosine had overall resistance of 5%, 1.7%, and 1.7%, respectively.
61.9 % were females and 38.2% were males, and 65.5% had low birth weight (<2.5 kg). Extremely low birth weight (≤1 kg) was significantly associated with candidemia (16.7%) compared to bacteremia (9%) (p<0.05). Prematurity was also prominent, with 65.5% of candidemia cases occurring in infants born before 36 weeks versus 47% in bacteremia (p<0.05). Notably, late preterm births (33-36 weeks) were significantly higher in candidemia (30.6%) than bacteremia (20.1%). Conversely, term births (>36 weeks) were significantly associated with bacterial BSIs (53% vs. 34.5%). These findings reaffirm preterm and low birth weight neonates as high-risk groups for candidemia12, 27.In our study, prior antibiotic exposure was documented in 71.4% of neonates with candidemia. Importantly, antibiotic use for less than one week was statistically significant (p<0.05) in candidemia (21.4%) compared with bacterial BSIs (11.5%). Our findings corroborate earlier studies by Fu et al.11, 12 and Benjamin et al., which demonstrated that broad-spectrum antibiotic exposure within seven days before diagnosis markedly increased candidemia risk in low-birth-weight neonates.
Ventilator support was required for 44% of the neonates with Candida BSI. Compared to neonatal bacteremia cases, which showed 13.2% of neonates requiring ventilatory assistance between 7 and 14 days, we found that 22.6% of neonatal candidemia patients required such ventilator support. This difference was statistically significant (p<0.05). Furthermore, ventilator support for longer than two weeks was linked to 13.1% of occurrences of newborn candidemia. This finding was statistically significant (p<0.05) in comparison to instances of newborn bacteremia, of which 6.8% required continuous ventilator support above 2 weeks.
In the present study, we observed several notable strengths in the form of identification of key risk factors such as low birth weight, prematurity, prior antibiotic exposure, and prolonged ventilator support adds important clinical relevance, further supported by statistical significance. However, certain limitations such as being a likely single-center observational study, the generalizability of the findings may be limited. Additionally, the absence of outcome measures such as mortality and lack of adjustment for potential confounders may influence interpretation.
CONCLUSIONS This study underscores the significance of neonatal bloodstream infections, with a predominance of non-albicans Candida species, particularly Candida tropicalis, and the emergence of antifungal resistance patterns in NICU settings. Prematurity, low birth weight, prior antibiotic exposure, and prolonged ventilator support were identified as the key risk factors for candidemia.
These findings emphasize the need for strict infection control measures, including adherence to hand hygiene and aseptic practices, along with implementation of robust antimicrobial stewardship programs to limit unnecessary antibiotic use. Routine surveillance of pathogen distribution and antifungal susceptibility, early identification of high-risk neonates with consideration of targeted prophylaxis, strengthening of laboratory diagnostics, and regular staff training are recommended to improve neonatal outcomes.
ACKNOWLEDGEMENT:
The technical help of Mr. Mohd Naved and Mr. Sanjay Sharma is duly acknowledged.
CONFLICT OF INTEREST STATEMENT:
No potential conflict of interest was reported by the authors.
SOURCE OF FUNDING:
None
REFERENCES
Odabasi IO, Bulbul A. Neonatal Sepsis. Sisli Etfal Hastan Tip Bul. 2020 Jun 12;54(2):142–58.
Nirmal K, Das S, Jothisri L, Jain C, Singh N pal. P367 Candida sepsis in neonates a neglected pathogen: study from neonatal intensive care unit tertiary care hospital. Med Mycol. 2022 Sep 20;60(Suppl 1):myac072P367.
Juyal D, Sharma M, Pal S, Rathaur VK, Sharma N. Emergence of Non-Albicans Candida Species in Neonatal Candidemia. N Am J Med Sci. 2013 Sep;5(9):541–5.
Lee Y, Puumala E, Robbins N, Cowen LE. Antifungal Drug Resistance: Molecular Mechanisms in Candida albicans and Beyond. Chem Rev. 2021 Mar 24;121(6):3390–411.
Berkow EL, Lockhart SR. Fluconazole resistance in Candida species: a current perspective. Infect Drug Resist. 2017 Jul 31;10:237–45.
Kelly MS, Benjamin DK, Smith PB. The Epidemiology and Diagnosis of Invasive Candidiasis Among Premature Infants. Clin Perinatol. 2015 Mar;42(1):105–17.
Rao S, Ali U. Systemic fungal infections in neonates. J Postgrad Med. 2005;51 Suppl 1:S27-29.
Zingg W, Tomaske M, Martin M. Risk of Parenteral Nutrition in Neonates—An Overview. Nutrients. 2012 Oct;4(10):1490–503.
Hamdan M, Puckett Y. Total Parenteral Nutrition. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 [cited 2023 Dec 5]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK559036/
Omran EA, Eisa FF, Bakr WMK. Microbial Contamination of Neonatal Injectable Lipid Emulsions at 12 and 24 Hours’ Infusion Time With Evaluation of Infection Control Measures. The Journal of Pediatric Pharmacology and Therapeutics : JPPT. 2020 Feb;25(1):53.
Benjamin DK, Stoll BJ, Gantz MG, Walsh MC, Sanchez PJ, Das A, et al. Neonatal Candidiasis: Epidemiology, Risk Factors, and Clinical Judgment. Pediatrics. 2010 Oct;126(4):e865–73.
Fu J, Ding Y, Jiang Y, Mo S, Xu S, Qin P. Persistent candidemia in very low birth weight neonates: risk factors and clinical significance. BMC Infectious Diseases. 2018 Nov 12;18(1):558.
Delaloye J, Calandra T. Invasive candidiasis as a cause of sepsis in the critically ill patient. Virulence. 2014 Jan 1;5(1):161–9.
Hope W, Natarajan P, Goodwin L. Invasive fungal infections. Clin Med (Lond). 2013 Oct;13(5):507–10.
Ly V, Sallam A. Fungal Endophthalmitis. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 [cited 2023 Dec 6]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK559257/
Bhattacharjee P. Epidemiology and antifungal susceptibility of Candida species in a tertiary care hospital, Kolkata, India. Curr Med Mycol. 2016 Jun;2(2):20–7.
Nazir A, Masoodi T. Spectrum of Candidal species isolated from neonates admitted in an Intensive Care Unit of teaching hospital of Kashmir, North India. J Lab Physicians. 2018;10(3):255–9.
Wayne PA. CLSI M44 Method for Antifungal Disk Diffusion Susceptibility Testing of Yeasts. 3rd ed. Pennsylvania, USA: Clinical and Laboratory Standards Institute; 2018. 44 p.
Biswas B, Sharma AK, Seema K, Kumar A, Boipai M, Kumar M. Emerging threat of Candida resistance among neonates at a teaching institute of Jharkhand. J Family Med Prim Care. 2023 May;12(5):946–52.
Tak V, Mathur P, Varghese P, Gunjiyal J, Xess I, Misra MC. The epidemiological profile of candidemia at an Indian trauma care center. J Lab Physicians. 2014 Jul;6(2):96–101.
Basu S, Kumar R, Tilak R, Kumar A. Candida Blood Stream Infection in Neonates: Experience from A Tertiary Care Teaching Hospital of Central India. Indian Pediatr. 2017 Jul 15;54(7):556–9.
Tunc G, Toksoz A, Kilicbay F. Candidal Infections in the Neonatal Intensive Care Unit: A Retrospective Observational Study. Sisli Etfal Hastan Tip Bul. 2023 Jun 20;57(2):204–9.
Roilides E, Farmaki E, Evdoridou J, Francesconi A, Kasai M, Filioti J, et al. Candida tropicalis in a Neonatal Intensive Care Unit: Epidemiologic and Molecular Analysis of an Outbreak of Infection with an Uncommon Neonatal Pathogen. J Clin Microbiol. 2003 Feb;41(2):735–41.
Xess I, Jain N, Hasan F, Mandal P, Banerjee U. Epidemiology of Candidemia in a Tertiary Care Centre of North India: 5-Year Study. Infection. 2007 Aug 1;35(4):256–9.
Gupta N, Mittal N, Sood P, Kumar S, Kaur R, Mathur M. CANDIDEMIA IN NEONATAL INTENSIVE CARE UNIT. Indian Journal of Pathology and Microbiology. 2001 Jan;44(1):45.
Kothari A, Sagar V. Epidemiology of Candida Bloodstream Infections in a Tertiary Care Institute in India. 2009 Oct 9;
Hsieh E, Smith PB, Benjamin DK. Neonatal fungal infections: when to treat? Early Hum Dev. 2012 May;88(Suppl 2):S6–10.
Submit a Manuscript:
Copyright © Author(s) 2026. JASPI- Journal of Antimicrobial Stewardship Practices and Infectious Diseases.

